FEATURED POST
What You Can Learn from 7 Theme Fusion Success Stories
Nam lacinia arcu tortor, nec luctus nibh dignissim eu. Nulla sit amet maximus nulla. Pellentesque a accumsan eros, ac molestie nulla. Morbi interdum in neque vitae vulputate.
Regulatory Compliance
Regulatory Compliance We not only know what to do but – how to do it. We ensure that your quality [...]
Lean Quality Solutions
Lean Quality Solutions for Medical Device & Pharmaceutical Companies Synergis, a sister company of BioTeknica, an IQVIA business, offers comprehensive [...]
BIOTEKNICA COMMUNITY OUTREACH
BIOTEKNICA COMMUNITY OUTREACH Peru, Miami, Africa! When not traveling around the world for client business, BioTeknica team members can be found [...]
ASR IS OVER! HOW DO VMSR & NEST IMPACT YOUR ORGANIZATION?
Feeling unsure of how the FDA’s updated reporting processes will affect you? You’re not alone. BioTeknica SMEs delved into [...]
KEY FDA EXPECTATIONS FOR ADVERSE EVENT REPORTING DURING A PANDEMIC
The FDA has modified the regulations regarding adverse event reporting during a pandemic to include COVID-19. Click below for [...]
BIOTEKNICA OPERATING AT FULL CAPACITY Company Implements Business Continuity Plan During COVID-19 Crisis
BioTeknica and its sister companies, Qualified Data Systems and Synergis, are here for you. We are open and operating [...]
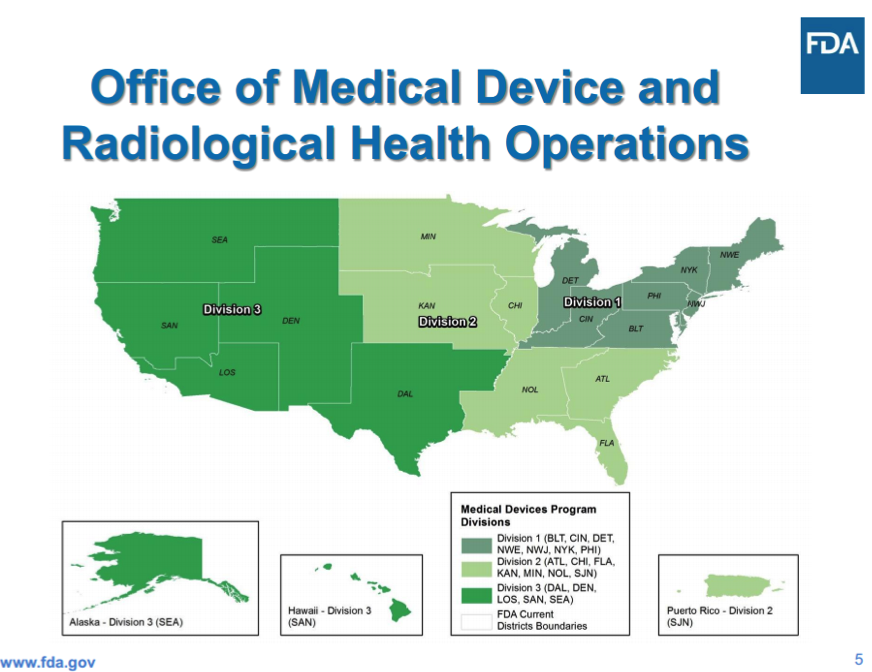
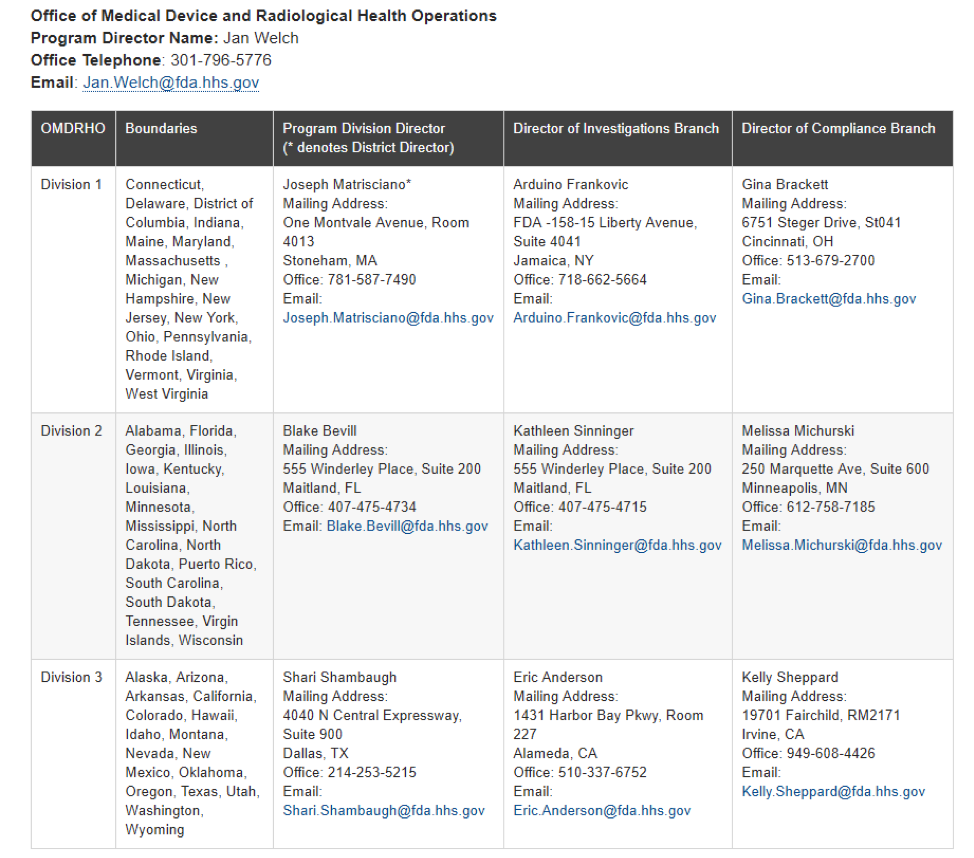
2 New Laws Impact Device Industry
2 New Laws Impact Device Industry According to a presentation by FDA official Blake Bevill, two recent laws [...]
Critical Issue Management
Critical Issue Management for FDA 483 Warning Letters, Recalls & Safety Alerts Agency Representation & [...]
Regulatory Compliance
Regulatory Compliance We not only know what to do but – how to do it. We [...]
Lean Quality Solutions
Lean Quality Solutions for Medical Device & Pharmaceutical Companies Synergis, a sister company of BioTeknica, an [...]
BIOTEKNICA COMMUNITY OUTREACH
BIOTEKNICA COMMUNITY OUTREACH Peru, Miami, Africa! When not traveling around the world for client business, BioTeknica team [...]
FDA’s Office Of Regulatory Affairs Realigned
FDA's Office Of Regulatory Affairs Realigned The FDA’s Office of Regulatory Affairs (ORA), the lead office for agency field activities, has changed its [...]
New FDA Communications Guidelines For Inspections
New FDA Communications Guidelines For Inspections Medical device companies have new FDA contacts and new guidelines to communicate with the FDA regarding their [...]
Modernizing the FDA Workforce
Modernizing the FDA Workforce A strategic priority identified by the FDA earlier this year is to strengthen the FDA’s scientific workforce by investing [...]
BioTeknica is an ISO 9001:2015 Certified Company
BioTeknica is an ISO 9001:2015 Certified Company BioTeknica has successfully achieved ISO 9001:2015 certification, the highest standards of its kind, for its Quality [...]
Lean Management Systems and the New Normal
Lean Management Systems and the New Normal Miguel E Guerrero, Lean Sensei, Managing Partner, Synergis Corp., a BioTeknica sister company The output of any [...]
FDA Program Alignment & The New CDRH in Bite-Sized Chunks
FDA Program Alignment & The New CDRH in Bite-Sized Chunks Changes at the FDA are impacting life science manufacturers. In 2013 the Agency [...]