FEATURED POST
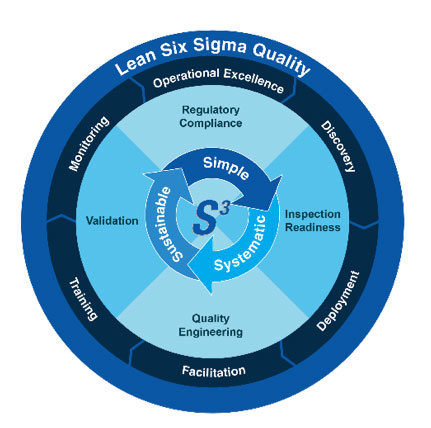
Quality Engineering
Are you able to resolve the root cause?
Quality Engineering services using proven methodologies to resolve life science engineering challenges
ASK A BIOTEKNICA SME HOW TO VALIDATE A PROCESS WITH A “HIGH DEGREE OF ASSURANCE” AND WHAT DOES THAT EVEN MEAN?
Looking for answers to your regulatory questions? To submit one, please click on Ask a BioTeknica SME and our Subject Matter Experts (SMEs) [...]
ASR IS OVER! HOW DO VMSR & NEST IMPACT YOUR ORGANIZATION?
Feeling unsure of how the FDA’s updated reporting processes will affect you? You’re not alone. BioTeknica SMEs delved into the real-world [...]
KEY FDA EXPECTATIONS FOR ADVERSE EVENT REPORTING DURING A PANDEMIC
The FDA has modified the regulations regarding adverse event reporting during a pandemic to include COVID-19. Click below for key points [...]
Get to the Head of the Class: Lunch & Learn Series at BioTeknica’s New Learning Center
Get to the Head of the Class: Lunch & Learn Series at BioTeknica's New Learning Center BioTeknica recently [...]
Julie Larsen Joins FDANews Medical Device Conference Advisory Board
Julie Larsen Joins FDANews Medical Device Conference Advisory Board Congratulations to BioTeknica principal Julie Larsen for her recent [...]
Charity: BioTeknica’s Helping Hands
Charity: BioTeknica's Helping Hands Every year for the past three and a half years, BioTeknica has partnered with Catholic Relief [...]
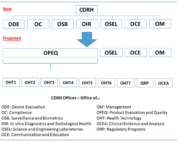
FDA Program Realignment: New Model for Device Approval & Inspection Efficiency
FDA Program Realignment: New Model for Device Approval & Inspection Efficiency The FDA Center for Devices and Radiological Health [...]
Operational Review
Operational Review & Optimization for Medical Device and Pharmaceutical Companies Cost Reduction Strategies Business Process Re-engineering (BPR) Process Optimization/Improvement Process Control and Automation [...]
ASK A BIOTEKNICA SME HOW TO VALIDATE A PROCESS WITH A “HIGH DEGREE OF ASSURANCE” AND WHAT DOES THAT EVEN MEAN?
Looking for answers to your regulatory questions? To submit one, please click on Ask a BioTeknica SME and our [...]
ASR IS OVER! HOW DO VMSR & NEST IMPACT YOUR ORGANIZATION?
Feeling unsure of how the FDA’s updated reporting processes will affect you? You’re not alone. BioTeknica SMEs [...]
KEY FDA EXPECTATIONS FOR ADVERSE EVENT REPORTING DURING A PANDEMIC
The FDA has modified the regulations regarding adverse event reporting during a pandemic to include COVID-19. Click [...]
FDA & EU Requirements and Expectations
5 Things You Should Know from the 2015 Association of Food & Drug Officials (AFDO) Conference If you didn’t have a chance to attend [...]
Successful Outcomes One Client at a Time
Successful Outcomes One Client at a Time Recently, BioTeknica assisted a leading medical device manufacturer to successfully address a previous FDA Warning Letter by [...]
Core Matters: Results that Yield Successful Business Outcomes & Substantial Compliance
We’re passionate about figuring out how to expertly integrate both the intent and interpretation of regulations to your specific products. So ask yourself how [...]