FEATURED POST
Quality Engineering
Are you able to resolve the root cause?
Quality Engineering services using proven methodologies to resolve life science engineering challenges
Regulatory Compliance
Regulatory Compliance We not only know what to do but – how to do it. We ensure that your quality [...]
Lean Quality Solutions
Lean Quality Solutions for Medical Device & Pharmaceutical Companies Synergis, a sister company of BioTeknica, an IQVIA business, offers comprehensive [...]
BIOTEKNICA COMMUNITY OUTREACH
BIOTEKNICA COMMUNITY OUTREACH Peru, Miami, Africa! When not traveling around the world for client business, BioTeknica team members can be found [...]
ASR IS OVER! HOW DO VMSR & NEST IMPACT YOUR ORGANIZATION?
Feeling unsure of how the FDA’s updated reporting processes will affect you? You’re not alone. BioTeknica SMEs delved into [...]
KEY FDA EXPECTATIONS FOR ADVERSE EVENT REPORTING DURING A PANDEMIC
The FDA has modified the regulations regarding adverse event reporting during a pandemic to include COVID-19. Click below for [...]
BIOTEKNICA OPERATING AT FULL CAPACITY Company Implements Business Continuity Plan During COVID-19 Crisis
BioTeknica and its sister companies, Qualified Data Systems and Synergis, are here for you. We are open and operating [...]
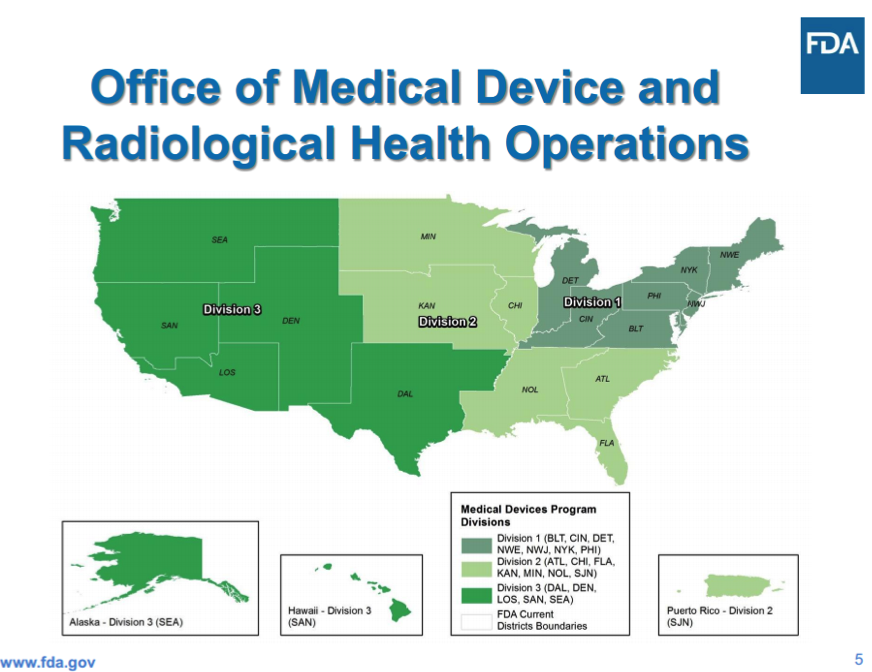
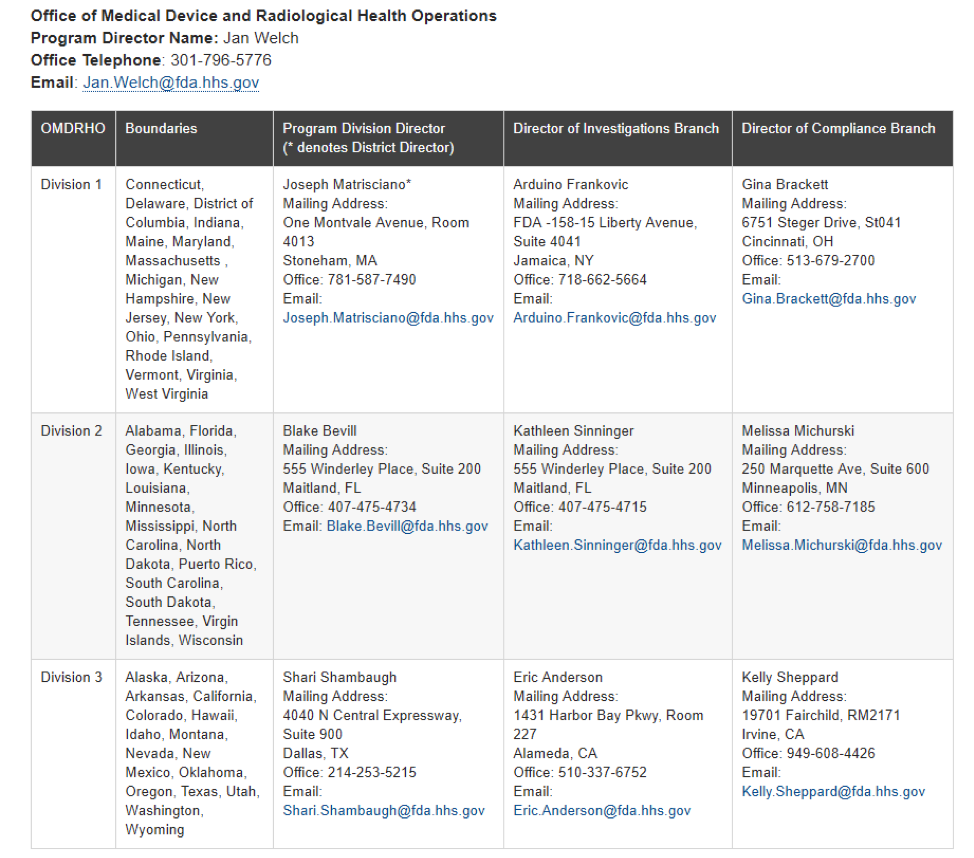
2 New Laws Impact Device Industry
2 New Laws Impact Device Industry According to a presentation by FDA official Blake Bevill, two recent laws [...]
Critical Issue Management
Critical Issue Management for FDA 483 Warning Letters, Recalls & Safety Alerts Agency Representation & [...]
Regulatory Compliance
Regulatory Compliance We not only know what to do but – how to do it. We [...]
Lean Quality Solutions
Lean Quality Solutions for Medical Device & Pharmaceutical Companies Synergis, a sister company of BioTeknica, an [...]
BIOTEKNICA COMMUNITY OUTREACH
BIOTEKNICA COMMUNITY OUTREACH Peru, Miami, Africa! When not traveling around the world for client business, BioTeknica team [...]
FDA’s Office Of Regulatory Affairs Realigned
FDA's Office Of Regulatory Affairs Realigned The FDA’s Office of Regulatory Affairs (ORA), the lead office for agency field activities, has changed its [...]
New FDA Communications Guidelines For Inspections
New FDA Communications Guidelines For Inspections Medical device companies have new FDA contacts and new guidelines to communicate with the FDA regarding their [...]
Modernizing the FDA Workforce
Modernizing the FDA Workforce A strategic priority identified by the FDA earlier this year is to strengthen the FDA’s scientific workforce by investing [...]
BioTeknica is an ISO 9001:2015 Certified Company
BioTeknica is an ISO 9001:2015 Certified Company BioTeknica has successfully achieved ISO 9001:2015 certification, the highest standards of its kind, for its Quality [...]
Lean Management Systems and the New Normal
Lean Management Systems and the New Normal Miguel E Guerrero, Lean Sensei, Managing Partner, Synergis Corp., a BioTeknica sister company The output of any [...]
FDA Program Alignment & The New CDRH in Bite-Sized Chunks
FDA Program Alignment & The New CDRH in Bite-Sized Chunks Changes at the FDA are impacting life science manufacturers. In 2013 the Agency [...]